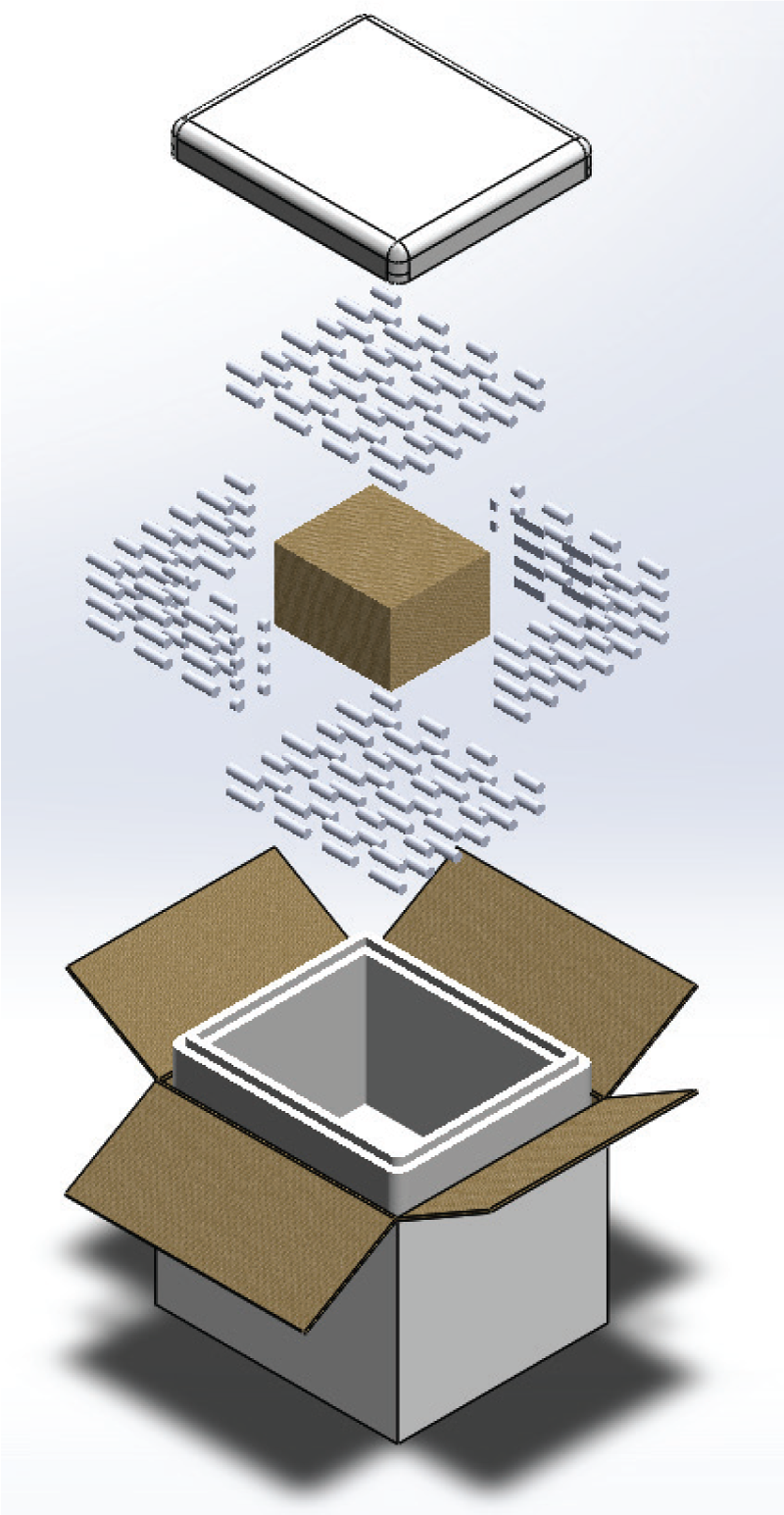
Seasonal transitions create inflection points across pharmaceutical supply chains, and vaccine distribution is no exception. As spring approaches, immunization programs often accelerate in response to public health initiatives, school requirements, international aid schedules, and post-winter inventory replenishment. These surges introduce higher shipment volumes, broader geographic reach, and more frequent handoffs, all while ambient temperatures begin to rise.
Vaccines are among the most temperature-sensitive biologics in circulation. Even minor deviations outside approved ranges can compromise potency and render doses ineffective. Preparing cold chains for spring distribution surges requires more than additional capacity. It demands systems engineered for repeatable thermal performance, design-tested packaging configurations, and logistics strategies that can absorb variability without increasing risk.
Seasonal Risk Factors That Strain Vaccine Cold Chains
Spring introduces a complex risk profile that differs significantly from winter distribution. While cold exposure dominates winter planning, transitional seasons challenge cold chains with fluctuating temperatures, longer transit windows, and mixed transport modes. Shipments may encounter cool morning conditions, warm afternoon exposure, and extended dwell times at transfer points, all within a single journey.
At the same time, spring distribution surges often coincide with expanded delivery networks. Vaccines may move beyond centralized healthcare facilities to community clinics, temporary immunization sites, and international destinations with inconsistent infrastructure. These conditions amplify the consequences of packaging or process failures.
Key seasonal stressors include:
- Rising ambient temperatures that increase heat gain during ground and air transport
- Higher shipment density that raises the likelihood of staging delays and cross-dock exposure
- Longer or more complex routes driven by expanded immunization programs
- Increased handling frequency across decentralized delivery points
Cold chain systems must be prepared to perform consistently across these variables, not only under ideal transport conditions.
Temperature Integrity as a Public Health Imperative
Vaccines rely on precise temperature control to preserve their biological structure and clinical effectiveness. Many formulations must remain within strict refrigerated or frozen ranges from manufacture through administration. Unlike many pharmaceutical products, temperature damage to vaccines is often invisible, meaning compromised doses may not be identified until efficacy is reduced in the field.
Maintaining temperature integrity is therefore not simply a regulatory requirement. It is fundamental to public trust in vaccination programs and the success of population-wide immunization efforts. During spring surges, when distribution volume increases and timelines compress, the margin for error narrows considerably.
Cold chain preparation must prioritize systems that deliver predictable performance without relying on manual intervention or excessive refrigerant. Thermal stability must be designed into the packaging itself, corroborated through testing that reflects real-world transit conditions rather than theoretical assumptions.
Design-Tested Packaging for Predictable Performance at Scale
Spring distribution surges expose the limitations of lightly qualified or improvised packaging solutions. To mitigate risk, vaccine shippers must be engineered and design-tested against standardized performance profiles that simulate seasonal variability.
Design testing to ISTA 7E protocols evaluates how packaging systems perform under both summer and winter temperature profiles, including transitional conditions common in spring. These tests assess insulation efficiency, refrigerant behavior, airflow dynamics, and payload interaction to confirm that internal temperatures remain within target ranges throughout transit.
Well-engineered vaccine shippers share several defining characteristics:
- Multi-component insulation systems that resist external temperature swings
- Optimized refrigerant placement that supports uniform internal temperatures
- Airflow designs that eliminate hot and cold zones within the payload space
- Confirmed duration profiles aligned with one-day, two-day, or extended transit needs
This level of qualification ensures that performance is repeatable across thousands of shipments, even as external conditions fluctuate.
Scaling Distribution Without Introducing Variability
As vaccine volumes increase in spring, cold chain strategies must scale without creating inconsistency. Packaging systems should be available in standardized formats that support multiple payload sizes and transit durations while maintaining identical performance characteristics.
Scalability depends on more than shipper availability. It requires manufacturing consistency and quality control processes that ensure each unit performs the same as the last. During surge periods, changes in materials, assembly methods, or refrigerant sourcing can introduce variability that undermines performance data established during testing.
Effective scale readiness is supported by:
- Standardized shipper configurations with documented pack-out procedures
- Multiple field-tested size options to match varying payload requirements
- Consistent material sourcing and controlled production processes
- Predictable lead times that support surge planning without last-minute substitutions
When packaging systems scale reliably, logistics teams can focus on execution rather than constant adjustment.
Monitoring and Operational Visibility During Peak Demand
 Temperature control does not end at shipment release. Monitoring and documentation provide essential visibility throughout transit, particularly during high-volume distribution periods. However, monitoring is most effective when paired with packaging systems designed to maintain stability independently.
Temperature control does not end at shipment release. Monitoring and documentation provide essential visibility throughout transit, particularly during high-volume distribution periods. However, monitoring is most effective when paired with packaging systems designed to maintain stability independently.
During spring surges, data should function as confirmation rather than constant alarm. When packaging is shown to withstand expected conditions, temperature monitoring supports compliance and audit readiness without overwhelming operations teams with avoidable alerts.
Preparation also includes clear training and documentation. Standardized assembly instructions, refrigerant conditioning guidelines, and handling protocols reduce variability across facilities and partners. These controls become increasingly important as temporary staff, third-party logistics providers, and new distribution points are introduced during seasonal expansion.
Supporting Diverse Distribution Environments
Spring vaccine distribution often extends beyond traditional healthcare channels. From school-based immunization programs to international aid deployments, cold chains must perform across a wide range of environments and infrastructure capabilities.
Packaging systems must therefore balance robustness with flexibility. Lightweight parcel shippers support last-mile delivery to clinics and pharmacies, while frozen configurations protect ultra-low temperature formulations during extended or international transport. The ability to deploy proven solutions across these use cases strengthens overall supply chain resilience.
This adaptability also supports cost control and sustainability objectives. Design-tested systems reduce the need for excessive refrigerant, minimize product loss, and streamline handling. Over time, these efficiencies contribute to lower total cost of ownership while reinforcing responsible cold chain practices.
Integrated Cold Chain Preparation for Seasonal Surges
Preparing vaccine cold chains for spring distribution surges is an ongoing process, not a one-time adjustment. It requires collaboration between packaging engineers, logistics planners, and quality teams to align solutions with evolving demand patterns.
Nordic Cold Chain Solutions supports this preparation through a comprehensive approach that combines design-tested packaging, on-site thermal testing, and expert consultation. From frozen and refrigerated vaccine shippers assessed to ISTA 7E standards to scalable configurations that support diverse routes and durations, these systems are engineered to perform under the pressures of seasonal expansion.
By investing in established cold chain readiness ahead of spring demand, pharmaceutical organizations can enter peak distribution periods with confidence. Consistent thermal performance, operational repeatability, and scalable design ensure that vaccines arrive effective, compliant, and ready to support the public health outcomes they are intended to deliver.